FDA Approves Endari, 1st Sickle Cell Treatment in Almost 20 Years, for Patients Age 5 and Older
shutterstock
Endari (L-glutamine) today became the first therapy in almost 20 years to win approval from the U.S. Food and Drug Administration to treat sickle cell disease.
In giving the green light to Endari use in patients starting at age 5, the FDA also made the oral drug the first ever approved to treat this blood disorder in children. Developed by Emmaus Life Sciences, Endari demonstrated in a pivotal clinical trial that it reduces severe complications associated with sickle cell disease, including hospitalizations for pain crises and occurrences of life-threatening acute chest syndrome.
“Until now, only one other drug was approved for patients living with this serious, debilitating condition,” Richard Pazdur, MD, acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research and director of the FDA’s Oncology Center of Excellence, said in a press release.
The therapy’s efficacy and safety were evaluated in the 48-week Phase 3 trial (NCT01179217) involving people with sickle cell disease between the ages of 5 and 58 who had two or more painful crises in the previous 12 months before enrollment. Researchers randomly assigned participants to receive Endari or a placebo.
On average, those treated with Endari had fewer hospital visits for pain treated with a narcotic administered by injection or infusion, or a ketorolac (a nonsteroidal anti-inflammatory drug), than participants who received placebo. They also had fewer hospitalizations for sickle cell pain, and spent fewer days in the hospital.
The Endari group also had fewer occurrences of acute chest syndrome, a serious complication of sickle cell disease, compared to the placebo group (8.6 percent vs. 23.1 percent).
Endari was found to be safe and generally well-tolerated, with common side effects including constipation, nausea, headache, abdominal pain, cough, and pain in the extremities, back, and chest.
The FDA’s Oncologic Drugs Advisory Committee recommended that Endari be approved to treat sickle cell disease in both adults and children on May 24. The regulatory agency previously had granted Endari orphan drug status, which provides incentives to assist and encourage the development of drugs for rare diseases.
Endari also has been designated an orphan drug in Europe.
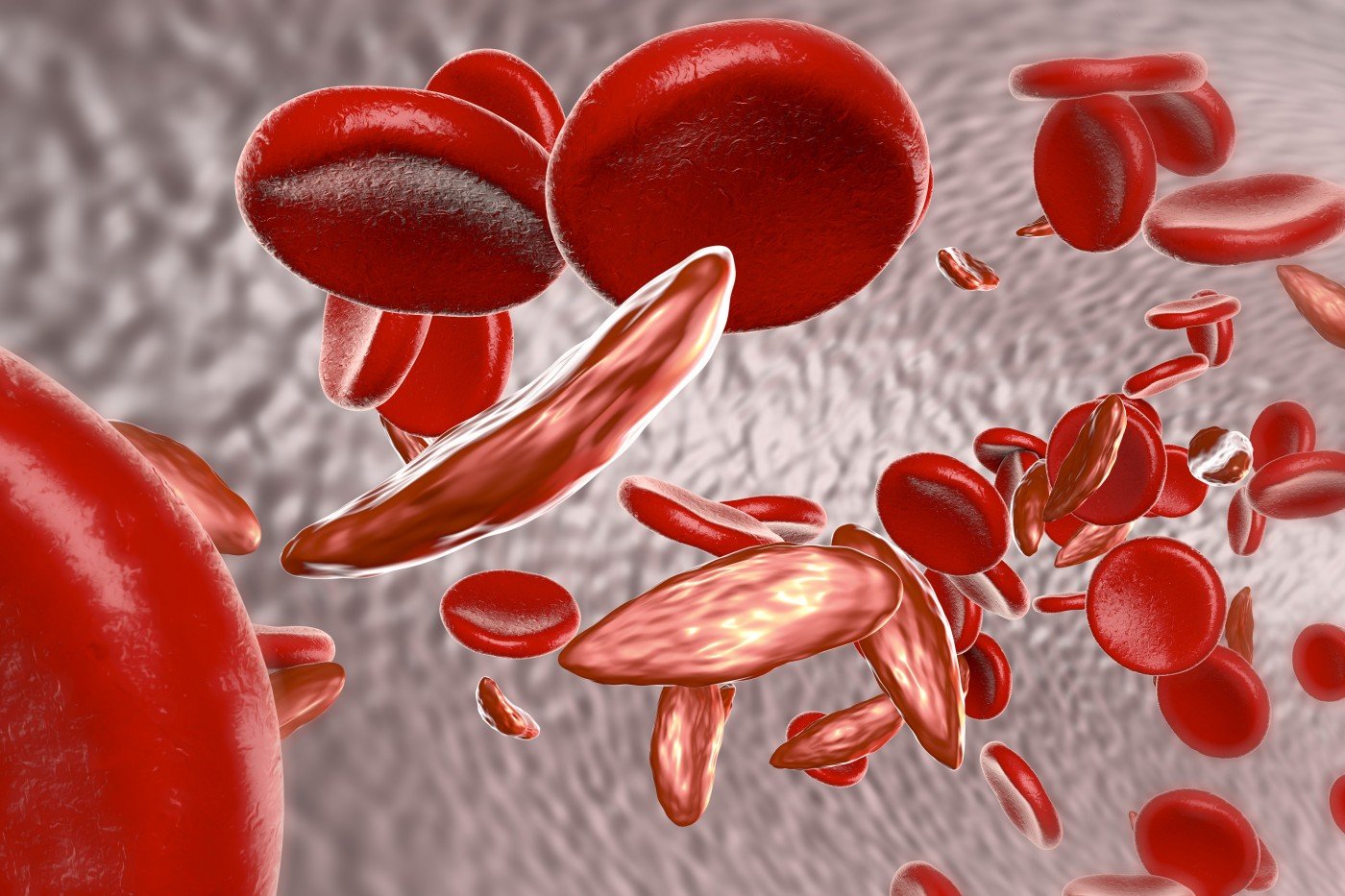
L-glutamine is an amino acid that increases the amount of free glutamine circulating in the blood. Sickle cell disease derives its name from the abnormally shaped red blood cells — resembling a crescent or sickle — that characterize this inherited blood disorder that affects an estimated 100,000 people in the U.S. The sickle cells often stick to blood vessel walls, blocking or slowing blood flow and oxygen transport.
The free glutamine is taken up by the sickle red blood cells and used to generate anti-oxidant molecules as a product of glutamine degradation. These new antioxidants help neutralize the oxidative stress in sickle cells, allowing them to regain the flexibility needed to travel through blood vessels and capillaries, carrying oxygen to tissues.